








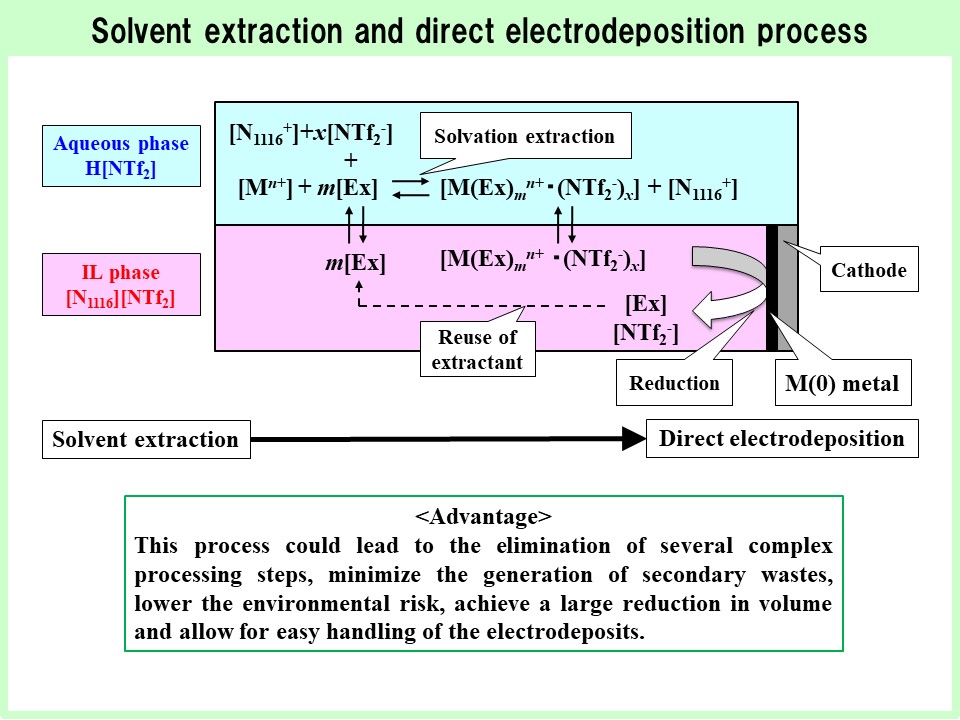
SX is a method for separating metal ions that utilizes the distribution equilibrium between the aqueous and organic phases. In our laboratory, we aim to develop efficient methods for separating rare metals; specifically, we are focusing on SX using IL system, which is characterized by the extraction of charged complexes. Furthermore, focusing particularly on PGM, we are developing SX-ED process that directly utilizes the IL including separated rare metals as the electrodeposition medium. This SX-ED process is effective from the perspective of reducing the volume of secondary waste. This research is being conducted as part of the 2023 and 2026 Grants-in-Aid for Scientific Research (B) projects. The analysis of the reduction and electrodeposition behaviors of extracted metal complexes is crucial. Therefore, our laboratory is conducting multifaceted and detailed analyses using high-precision combined electrochemical measurement methods, such as the electrochemical quartz crystal microbalance (EQCM) method, microelectrode measurements, scanning electrochemical microscopy (SECM), and photoelectrochemical measurements.
・PGM----platinum group metals
・SAC----spent automobile catalyst
・SX----solvent extraction
・ED----electrodeposition
・IL----ionic liquids
※Research results
・M. Matsumiya, S. Tokumitsu, T. Mishima, Y. Sasaki, ECS Adv., 3 (2024) 043001.
・R. Kinoshita, Y. Sasaki, M. Kaneko, M. Matsumiya, K. Shinoku, H. Shiroishi, Hydrometallurgy, 222 (2023) 1575-1583.
・Y. Sasaki, M. Kaneko, Y. Ban, R. Kinoshita, M. Matsumiya, K. Shinoku, H. Shiroishi, Anal. Sci., 39(9) (2023) 106159.
・M. Matsumiya, R. Kinoshita, Y. Sasaki, J. Electrochem. Soc., 169 (2022) 082513.
・R. Kinoshita, M. Matsumiya, Y. Sasaki, Solvent Extr. Ion Exch.,40(6) (2022) 606-619.
・M. Matsumiya, Y. Tsuchida, R. Kinoshita, Y. Sasaki, J. Electrochem. Soc., 168(7) (2022) 076508.
・M. Matsumiya, R. Kinoshita, Y. Tsuchida, Y. Sasaki, J. Electrochem. Soc., 168(5) (2022) 056501.
・M. Matsumiya, Y. Song, Y. Tsuchida, Y. Sasaki, Sep. Purif. Technol., 234 (2020) 115841.
・M. Matsumiya, Y. Song, Y. Tsuchida, H. Ota, K. Tsunashima, Sep. Purif. Technol., 214 (2019) 162-167.
・Y. Song, Y. Tsuchida, M. Matsumiya, K. Tsunashima, Hydrometallurgy, 181 (2018) 164-168.
・M. Matsumiya, M. Sumi, Y. Uchino, I. Yanagi, Sep. Purif. Technol., 201 (2018) 25-29.
・M. Matsumiya, T. Yamada, Y. Kikuchi, S. Kawakami, Solvent Extr. Ion Exch., 34(6) (2016) 522-534.
・M. Matsumiya, T. Yamada, S. Murakami, Y. Kohno, K. Tsunashima, Solvent Extr. Ion Exch., 34(5) (2016) 454-468.
・M. Matsumiya, Y. Kikuchi, T. Yamada, S. Kawakami, Sep. Purif. Technol., 130 (2014) 91-101.
※Research grant
・Grant-in-Aid for Scientific Research (B) (26K01541)
・Grant-in-Aid for Scientific Research (B) (23H02002)
・JST Adaptable and Science Technology transfer Program (A-STEP) (MP27115663745)
・Grant-in-Aid for Exploratory Research (15K14193)
・Sasakawa Scientific Research Grant, Nanae Tsuda (25-313)
・JST Adaptable and Science Technology transfer Program (A-STEP) (AS242Z02857M)
・JST Adaptable and Science Technology transfer Program (A-STEP) (AS232Z01827C)
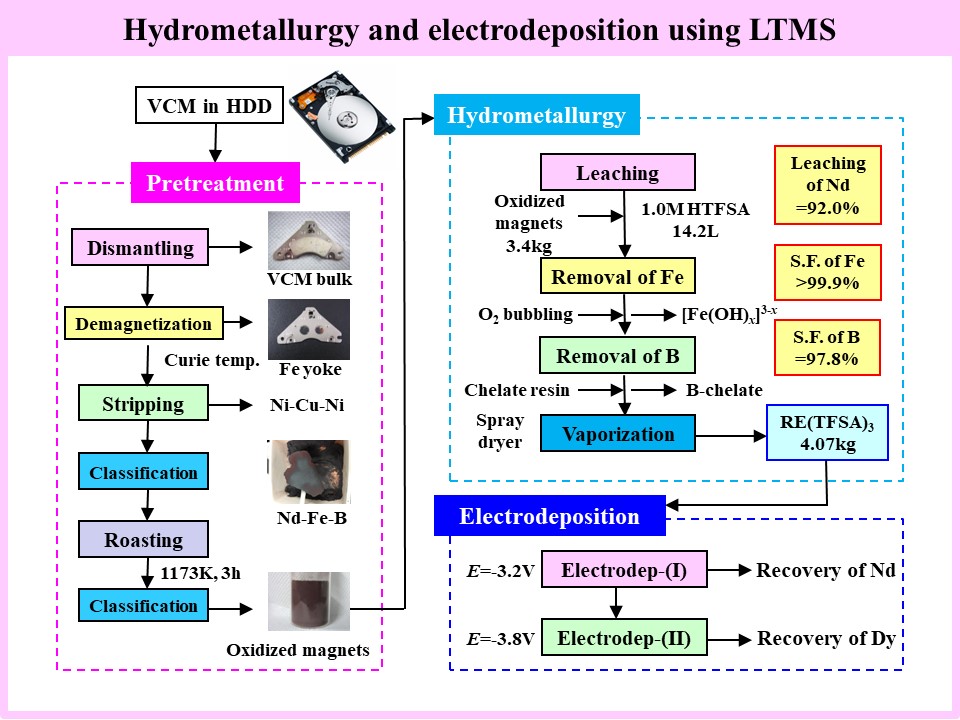
In recent years, measures to ensure a stable supply of RE resources have been prioritized at the national level, and there is a growing need to develop Japan’s proprietary recovering technologies for REE to reduce dependence on other countries. Based on our accumulated research on ED in IL systems, our laboratory has demonstrated a process for efficiently recovering REE from actual wastes, specifically, Nd-Fe-B magnets using low-temperature molten salts (LTMS) composed of anionic components of ILs. Dysprosium (Dy), in particular, has high resource value, making its selective separation crucial. The ED process using LTMS is an energy-saving, low-temperature technology that efficiently recovers Dy metal. Furthermore, through joint research with the Japan Atomic Energy Agency (JAEA), we are developing a process for the mutual separation of Nd and Dy by introducing a multi-stage SX using diglycolamide (DGA)-type extractants, which exhibit high REE separation performance, in hydrometallurgy.
・REE----rare earth elements
・LTMS----low-temperature molten salts
・ED----electrodeposition
・IL----ionic liquids
※Research results
・D. Nomizu, Y. Sasaki, M. Kaneko, M. Matsumiya, S. Katsuta, J. Radioanal. Nucl. Chem., 331 (2022) 1483-1493.
・M. Matsumiya, D. Nomizu, Y. Tsuchida, Y. Sasaki, J. Electrochem. Soc., 168(5) (2022) 056502.
・Y. Sasaki, M. Matsumiya, Y. Tsuchida, Anal. Sci., 36(11) (2020) 1303-1309.
・Y. Sasaki, Y. Ban, K. Morita, M. Matsumiya, R. Ono, H. Shiroishi, SERDJ, 27(1) (2020) 63-67.
・S. Murakami, M. Matsumiya, Y. Sasaki, S. Suzuki, S. Hisamatsu, K. Takao, Solvent Extr. Ion Exch., 35(4) (2017) 233-250.
・M. Matsumiya, H. Ota, K. Kuribara, K. Tsunashima, J. Electrochem. Soc., 164(8) (2017) H5230-H5235.
・H. Ota, M. Matsumiya, T. Yamada, T. Fujita, S. Kawakami, Sep. Purif. Technol., 170 (2016) 417-426.
・S. Murakami, M. Matsumiya, T. Yamada, K. Tsunashima, Solvent Extr. Ion Exch., 34(2) (2016) 172-187.
・M. Matsumiya, M. Ishii, R. Kazama, S. Kawakami, Electrochim. Acta, 146 (2014) 371-377.
・K. Ishioka, M. Matsumiya, M. Ishii, S. Kawakami, Hydrometallurgy, 144-145 (2014) 186-194.
・R. Kazama, M. Matsumiya, N. Tsuda, K. Tsunashima, Electrochim. Acta, 113 (2013) 269-279.
・A. Kurachi, M. Matsumiya, K. Tsunashima, S. Kodama, J. App. Electrochem., 42(11) (2012) 961-968.
・H. Kondo, M. Matsumiya, K. Tsunashima, S. Kodama, Electrochim. Acta., 66(1) (2012) 313-319.
※Research grant
・JST Adaptable and Science Technology transfer Program (A-STEP) (JPMJTM22C1)
・JST Adaptable and Science Technology transfer Program (A-STEP) (JPMJTM20D6)
・Sasakawa Scientific Research Grant, Daiki Nomizu (2021-3040)
・Grant-in-Aid for Scientific Research (B) (18H03404)
・JST Adaptable and Science Technology transfer Program (A-STEP) (VP29117940881)
・Grant-in-Aid for Scientific Research (B) (15H02848)
・Sasakawa Scientific Research Grant, Naoko Sasaya (27-334)
・JST Adaptable and Science Technology transfer Program (A-STEP) (AS2621394M)
・Grant-in-Aid for Young Scientists (A) (24686101)
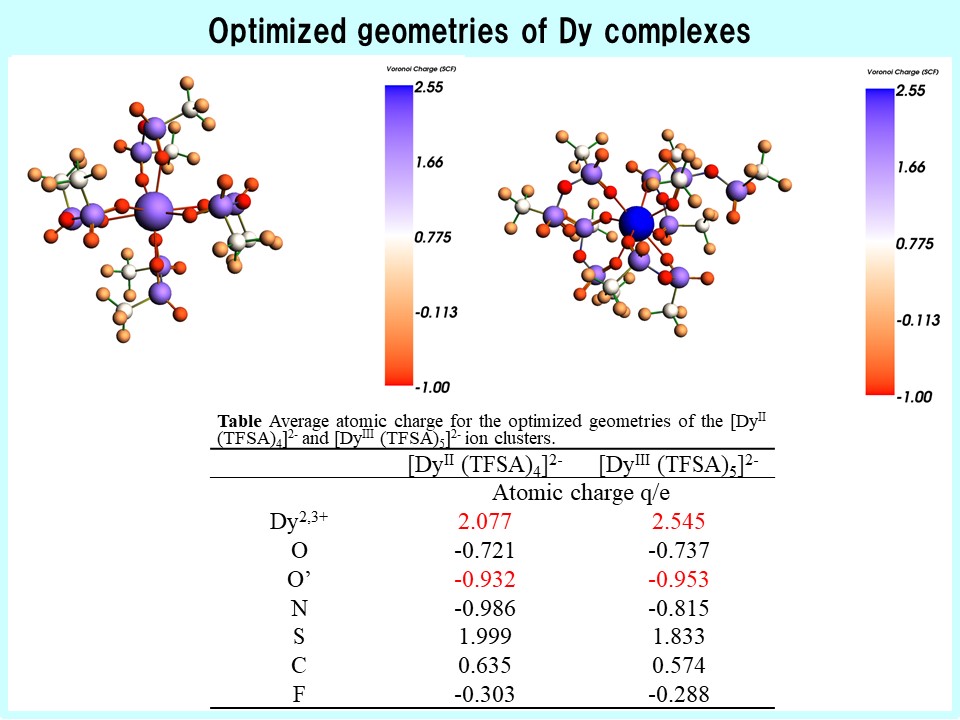
Due to their low vapor pressure, low environmental diffusivity, and inflame properties, ILs are expected to find applications as chemical reaction media and in electrochemical devices. The bulk properties of these ILs are determined by the motion of ions within the liquid. This ionic motion is governed by interionic interactions. Therefore, to elucidate the correlation between the microscopic structure and macroscopic physical properties of ILs, information regarding interionic interactions and ionic motion is essential. In particular, during the ED of rare metals, static structures such as the solvation structure of metal ions and dynamic behaviors such as diffusion coefficients are critical factors. Therefore, our laboratory evaluates the solvation structures of metal complexes through a combined analysis of spectroscopic methods, such as Raman spectroscopy and DFT calculations.
・DFT----density functional theory
・ED----electrodeposition
・IL----ionic liquids
※Research results
※Research grant
・D. Nomizu, Y. Tsuchida, M. Matsumiya, K. Tsunashima, J. Mol. Liq., 318 (2020) 114008.
・Y. Tsuchida, M. Matsumiya, K. Tsunashima, J. Mol. Liq., 269 (2018) 8-13.
・M. Matsumiya, R. Kazama, K. Tsunashima, J. Appl. Sol. Chem. Model., 5(4) (2016) 157-167.
・M. Matsumiya, R. Kazama, K. Tsunashima, J. Mol. Liq., 215 (2015) 308-315.
・M. Matsumiya, K. Hata, K. Tsunashima, J. Mol. Liq., 203 (2015) 125-130.
・M. Matsumiya, Y. Kamo, K. Hata, K. Tsunashima, J. Mol. Struct., 1048 (2013) 59-63.
・Sasakawa Scientific Research Grant, Ryoma Kinoshita (2022-2027)
・Sasakawa Scientific Research Grant, Ryo Kazama (26-216)